‘Explosive’ Spike In COVID-19 Cases Continues In Dallas County
As Dallas County continues to see an “explosive spike in COVID-19 cases and hospitalizations and there’s more hopeful news on vaccine candidates, here’s what you need to know today:
- Dallas County reports 1,831 new COVID-19 cases, one more death;
- Early data shows Moderna’s COVID-19 vaccine 94.5% effective;
- Small business grant program to give up to $3,000 to eligible small businesses, nonprofits.
Dallas County Reports 1,831 New COVID-19 Cases, One More Death
Dallas County Health and Human Services Monday reported 1,831 more positive cases of COVID-19 – 1,695 confirmed cases and 136 probable – and one additional death.
The county’s reported a cumulative total of 110,717 cases and 1,143 confirmed deaths, as well as 9,998 probable cases and 20 probable deaths.
The additional death reported Monday was a man in his 80s who lived in a Dallas long-term care facility. He didn’t have underlying conditions.
Of the total confirmed deaths reported to date, about 24% have been associated with long-term care facilities. Of all confirmed cases requiring hospitalization to date, more than two-thirds have been under 65 years of age.
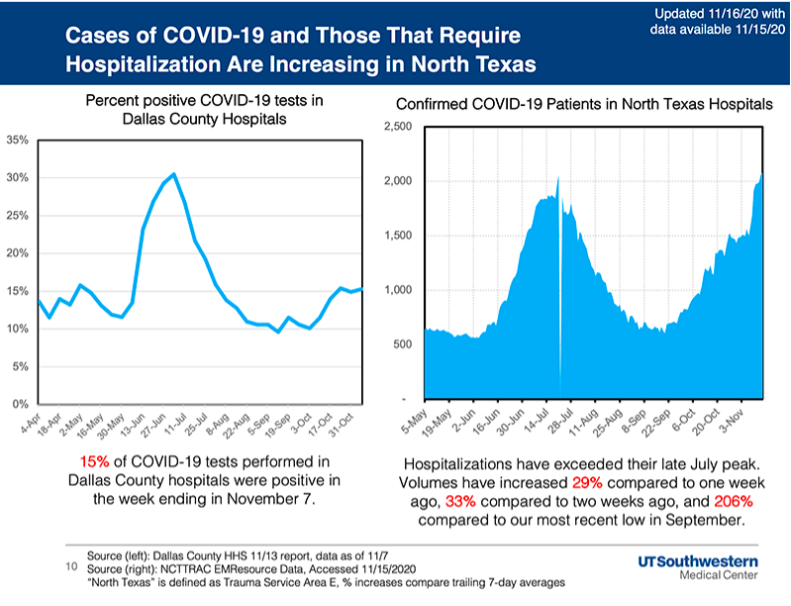
The percentage of respiratory specimens testing positive for the virus that causes COVID-19 also remains high, the county says.with 15.3% of symptomatic patients presenting to area hospitals testing positive in the week ending Nov. 7.
“In the last three days, we have seen almost 5,000 cases. This is indicative of the explosive nature of the spike that we are now in and it is imperative to public health and our economy that we stop the in-home get-togethers and trips to restaurants and bars that are largely responsible for this spike,” said Dallas County Judge Clay Jenkins. “Please do your part by sacrificing your understandable desire to do the things that the doctors tell us are just not safe right now. Focus on doing your duty to protect your family and your community. COVID will not be with us forever and there is good news with vaccines that will be here soon, but it is with us now even though we are tired and wish it were not. It is incumbent on all of us as patriotic Americans and North Texans to do our part to stop the spread of this dangerous disease.”
A provisional total of 843 confirmed and probable COVID-19 cases were diagnosed in school-aged children (5 to 17 years) during the week ending Nov. 7 and, during this past week, 10 K-12 schools in Dallas County have initiated temporary closures of their campuses to in-person instruction due to COVID-19 cases.
As of Monday, Highland Park ISD reported one case among a staff member assigned to the Armstrong Elementary campus, one in a student there, one case among a student at Boone Elementary, one among a staff member assigned to Bradfield, one case among a staff member assigned to Hyer, one case among a student there, three cases among students at University Park, two cases among staff members assigned to McCulloch Intermediate, three among students there, one case among a staff member assigned to Highland Park Middle School, two among students there, and five cases among students at Highland Park High School.
As of Monday, Dallas ISD reported 1,066 cases districtwide, 437 among campus staff, 107 among central staff, and 522 among students.
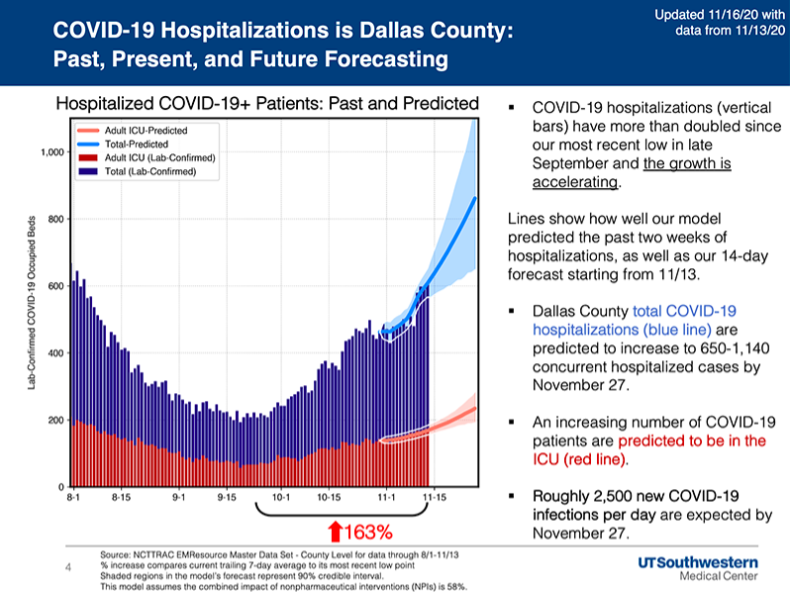
UT Southwestern Medical Center also reports hospitalizations in North Texas surpassed the previous peak levels of late July and the average volume for the last week was 206% higher than the most recent low in late September.
UTSW’s model projects total COVID-19 infections in Dallas County could increase to between 650 and 1,140 concurrent hospitalized cases by Nov. 27 and roughly 2,500 new COVID-19 infections per day are expected by Nov. 27.
Early Data Shows Moderna’s COVID-19 Vaccine 94.5% Effective

Biotechnology company Moderna announced Monday that their own COVID-19 vaccine candidate appeared to be 94.5% effective, according to early data from an ongoing study.
Moderna, like Pfizer, which recently reported their vaccine candidate appears to be 90% effective per early data, plans to submit for an emergency use authorization (EUA) with the U.S. Food and Drug Administration (FDA) in the coming weeks. Moderna also plans to submit applications for authorizations to global regulatory agencies.
“This is a pivotal moment in the development of our COVID-19 vaccine candidate. Since early January, we have chased this virus with the intent to protect as many people around the world as possible. All along, we have known that each day matters. This positive interim analysis from our Phase 3 study has given us the first clinical validation that our vaccine can prevent COVID-19 disease, including severe disease,” said Stéphane Bancel, CEO of Moderna, in a statement.
Both vaccine candidates require two shots.
Moderna says they expect to have about 20 million doses ready to ship in the U.S. by the end of the year and remains on track to manufacture 500 million to 1 billion doses globally in 2021.
Pfizer estimated it could produce globally up to 50 million vaccine doses in 2020 and up to 1.3 billion doses in 2021.
CVS Health shared in an email that they will receive a supply of the COVID-19 vaccine when it’s authorized and available to administer in pharmacies nationwide.
“We are proud of and thankful for the efforts of our pharmacists, nurse practitioners, and pharmacy technicians. They have served many communities across the U.S. and helped conduct nearly 7 million COVID-19 tests. We know they stand ready to do the same thing when COVID-19 vaccinations are available,” the company shared in the email.
Small Business Grant Program To Give Up To $3,000 To Eligible Small Businesses, Nonprofits
B.U.I.L.D. (Broaden Urban Investments to Leverage Dallas) has launched the B.U.I.L.D small business grant program to assist small businesses impacted by the economic downturn caused by the global COVID-19 pandemic. The program will provide businesses in the city of Dallas that have fewer than nine employees, or a 2019 payroll of less than $250,000, with a one-time grant of up to $3,000.
The primary focus of the program is to fund minority women-owned businesses in southern Dallas.
The grants can be used for payroll, rent, utilities, inventory, purchase of machinery, or equipment.
Applications are now open and will close on Nov. 24. Funds are anticipated to be disbursed by December 30. Information on eligibility requirements, restrictions, and other aspects of the program are published on the B.U.I.L.D. grant portal.





